|
1/4/2023 0 Comments Molecular geometry 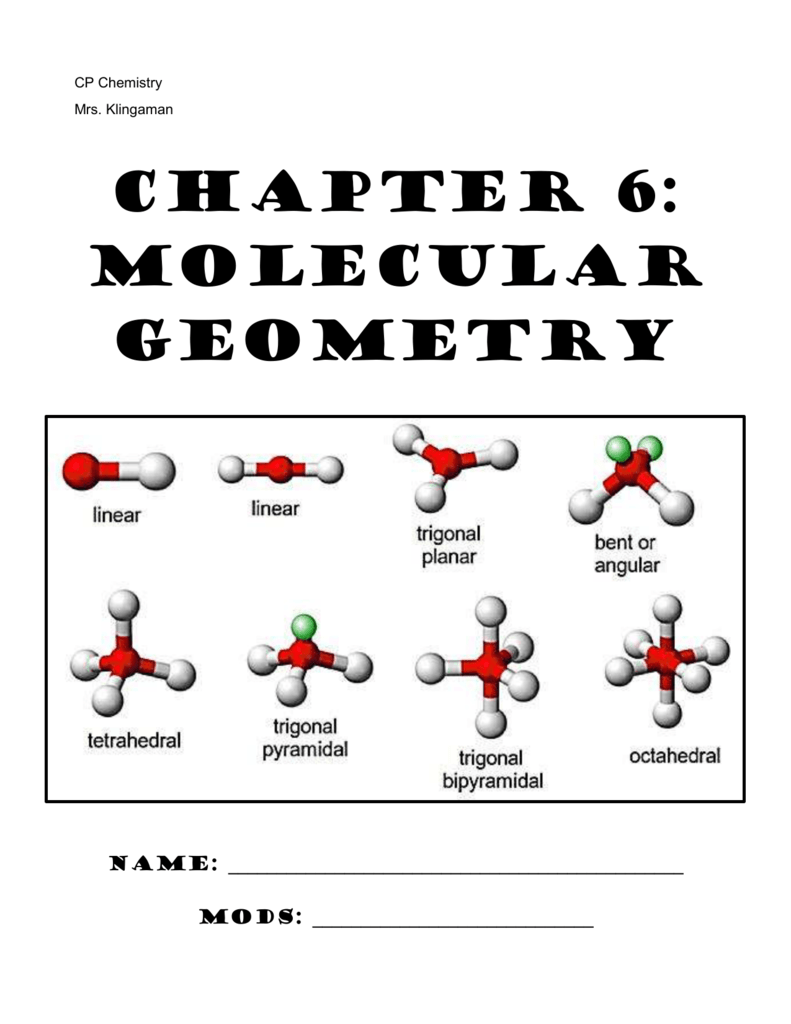
H 2O is an example of a molecule with a central atom that has four electron groups but only two of them are bonded to surrounding atoms.Īlthough the electron groups are oriented in the shape of a tetrahedron, the shape of the molecule is bent or angular. NH 3 is an example of a molecule whose central atom has four electron groups but only three of them are bonded to surrounding atoms.Īlthough the electron groups are oriented in the shape of a tetrahedron, from a molecular geometry perspective, the shape of NH 3 is trigonal pyramidal. The straight lines are in the plane of the page, the solid wedged line is coming out of the plane toward the reader, and the dashed wedged line is going out of the plane away from the reader. This diagram of CH 4 illustrates the standard convention of displaying a three-dimensional molecule on a two-dimensional surface. Figure 9.4 “Tetrahedral Geometry.” Four electron groups orient themselves in the shape of a tetrahedron. This shape is called bent or angular.Ī molecule with four electron groups around the central atom orients the four groups in the direction of a tetrahedron, as shown in Figure 9.4 “Tetrahedral Geometry.” If there are four atoms attached to these electron groups, then the molecular shape is also tetrahedral. An example is GeF 2:įrom an electron-group-geometry perspective, GeF 2 has a trigonal planar shape, but its real shape is dictated by the positions of the atoms. Some substances have a trigonal planar electron group distribution but have atoms bonded to only two of the three electron groups. The shape of such molecules is trigonal planar. They adopt the positions of an equilateral triangle - 120° apart and in a plane. Examples include BeH 2 and CO 2:Ī molecule with three electron groups orients the three groups as far apart as possible.  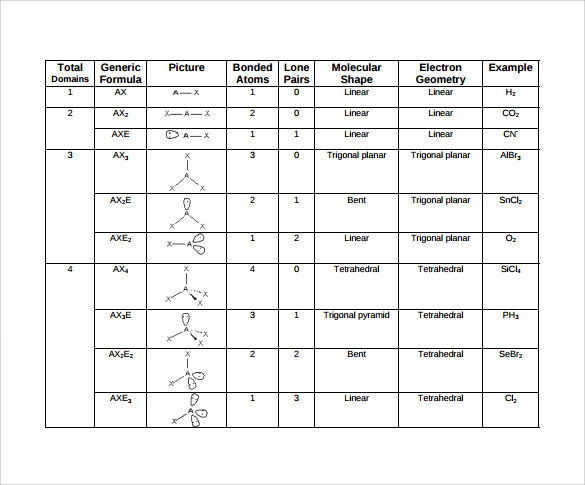
When the two electron groups are 180° apart, the atoms attached to those electron groups are also 180° apart, so the overall molecular shape is linear. A molecule whose central atom contains only two electron groups orients those two groups as far apart from each other as possible - 180° apart. Remember that a multiple bond counts as only one electron group.Īny molecule with only two atoms is linear. When applying VSEPR to simple molecules, the first thing to do is to count the number of electron groups around the central atom. There are two types of electron groups: any type of bond - single, double, or triple - and lone electron pairs. VSEPR makes a distinction between electron group geometry, which expresses how electron groups (bonding and nonbonding electron pairs) are arranged, and molecular geometry, which expresses how the atoms in a molecule are arranged. It says that electron pairs, being composed of negatively charged particles, repel each other to get as far away from each other as possible. The basic idea in molecular shapes is called valence shell electron pair repulsion (VSEPR). Small molecules - molecules with a single central atom - have shapes that can be easily predicted. There is an abundance of experimental evidence to that effect - from their physical properties to their chemical reactivity. Determine the polarity of molecules using net molecular dipoles.Determine the shape of simple molecules. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
